|
5/4/2023 0 Comments Abc transporter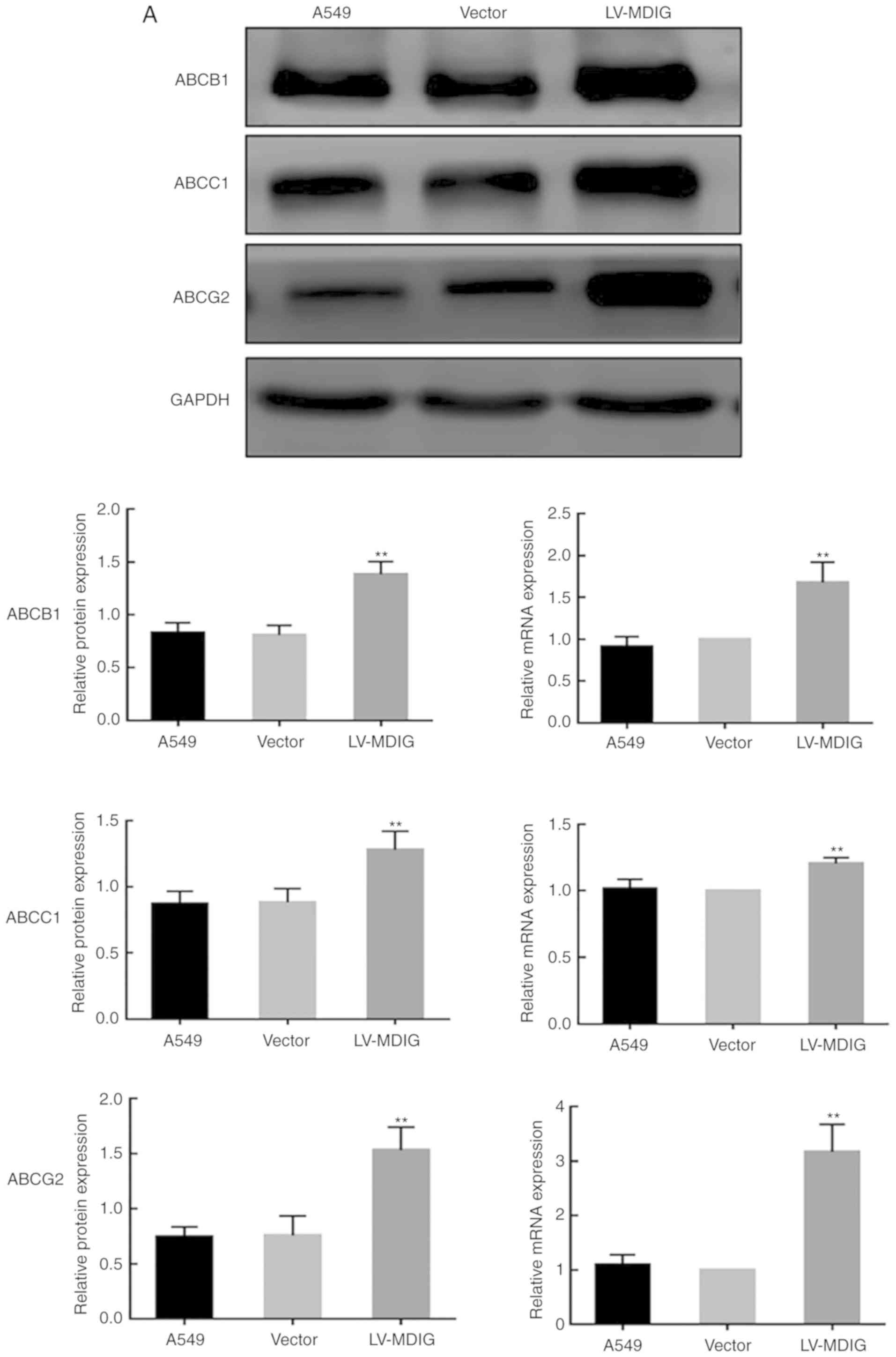
In vitro studies with HeLa cell line sensitive and resistant to actinomycin D. Multidrug resistance in cancer: role of ATP-dependent transporters. Mammalian drug efflux transporters of the ATP binding cassette (ABC) family: an overview. ABC multidrug transporters: structure, function and role in chemoresistance. The controversial role of ABC transporters in clinical oncology. Tamaki, A., Ierano, C., Szakacs, G., Robey, R. Toward a better understanding of the complexity of cancer drug resistance. With this Opinion article, we present recent evidence indicating that it is time to revisit the investigation into the role of ABC transporters in efficient drug delivery in various cancer types and at the blood–brain barrier. However, with the emergence of new data and advanced techniques, we propose to re-evaluate whether these transporters play a clinical role in multidrug resistance. Thirty years after the initial cloning and characterization of MDR1 and the implication of two additional ABC transporters, the multidrug resistance-associated protein 1 (MRP1 encoded by ABCC1)), and ABCG2, in multidrug resistance, interest in investigating these transporters as therapeutic targets has waned. At the time of its discovery, appropriate tools for the characterization and clinical development of MDR1 as a therapeutic target were lacking. However, the development of MDR1 as a therapeutic target has been unsuccessful. Ample evidence suggests that the expression of ATP-binding cassette (ABC) transporters, especially the multidrug resistance protein 1 (MDR1, also known as P-glycoprotein or P-gp), which is encoded by ABC subfamily B member 1 ( ABCB1), can confer resistance to cytotoxic and targeted chemotherapy. We recently characterized the binding site of propaphenon like substrate and found that substrate can take two distinct routes while moving through the transporter.Most patients who die of cancer have disseminated disease that has become resistant to multiple therapeutic modalities. Clarke, resulting in a model that is in line with all experimental data. We developed a model of the P-glycoprotein in the ATP-bound state and modified the model conformation by making extensive use of experimental cross-links determined by the group of D. This procedure might have influenced the conformation of transporter and lead to the structures observed in the crystals. During crystallization, the transporter is extracted from its native membrane environment and solubilized by surfactants. Some uncertainty with respect to the physiological conformation of the transporter remained, as the structures and models are typically not fully in line with all biochemical evidence. Model of the transporterĬrystallization of ABC exporter prepared the ground for modeling P-glycoprotein. Structural differences corresponding to the high and low affinity states for substrates, the occluded state as well as solute uptake from the membrane have to be defined yet. 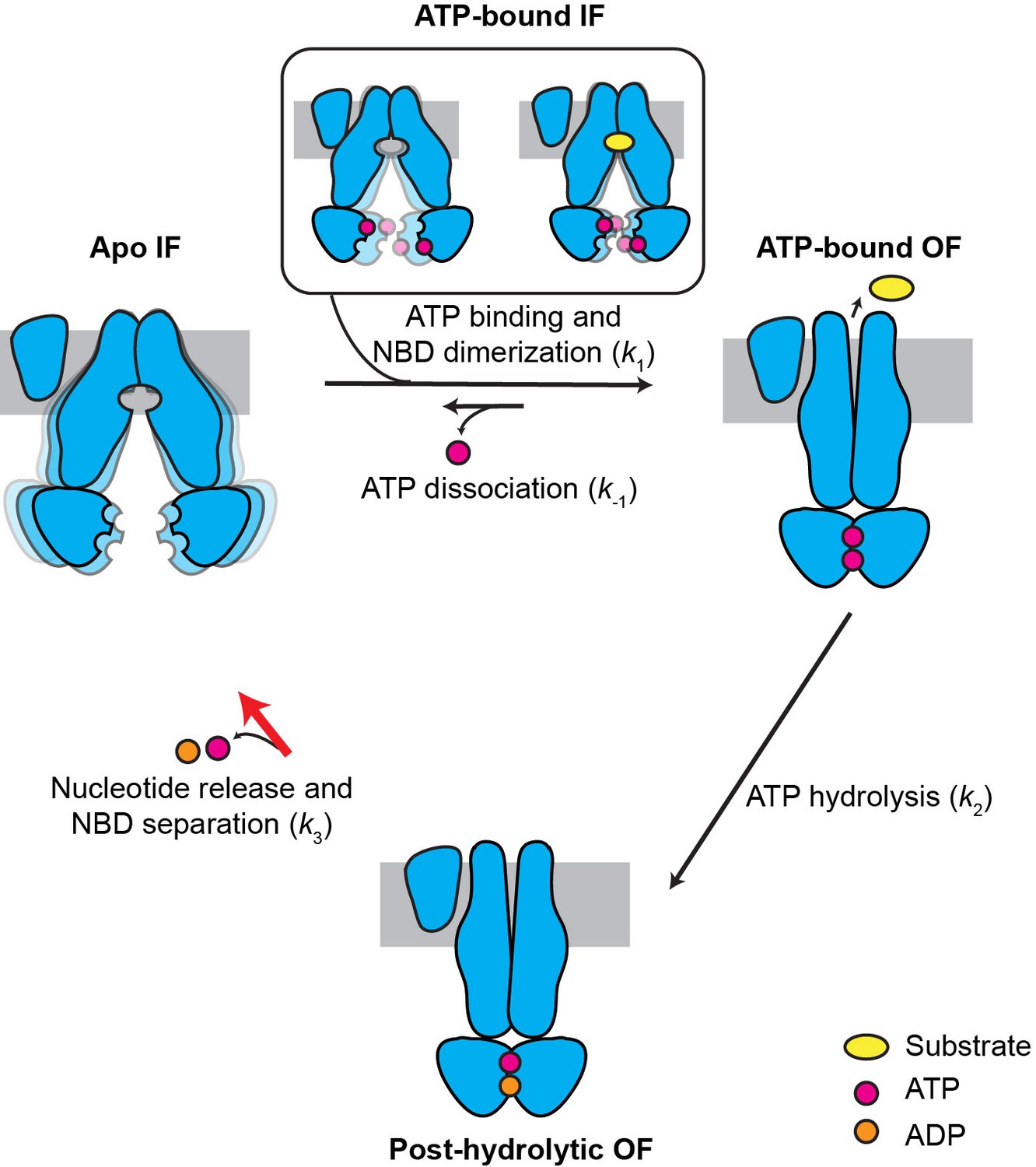
A molecular detailed transport model would be highly desirable. They agree in that ATP-binding and hydrolysis powers the transport cycle and that the conformational changes in the NBDs are transmitted to the TMDs, inducing a conformational transition from the substrate-binding inward-facing to the substrate-releasing outward=facing conformation. Several conceptual substrate transport models have been developed. The second ABC transporter in our focus of interest is the bile salt transporter (BSEP, ABCB11), which shares 49 % sequence identity with P-glycoprotein, but shows a very narrow substrate spectrum, limited essentially to bile salts. P-glycoprotein or breast cancer resistance protein (the second defence ABC transporter) are often upregulated in cancer cells, where they alone or in tandem confer resistance to chemotherapy. P-glycoprotein is known to intercept thousands of different, often structurally unrelated, hydrophobic compounds within the cell membrane. It is expressed at barrier tissues including kidney, intestine, blood-brain-barrier, placenta, as well as liver. We focus on P-glycoprotein (ABCB1, P-gp), which is a defence system evolved to prevent xenophobic substances form entering our body. accessible through molecular dynamics simulations. The full transport cycle will require characterization of the dynamics, as e.g. A lot of structural information is now available for the ABCB subfamily, as crystal structure of several member have now been solved. Common to all ABC transporter are the nucleotide binding domains, which provide the driving force by hydrolising ATP. Most members are membrane exporters, while four are involved in gene expression regulation. The human genome codes for 48 members of the large family of ABC proteins.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
